Adare Pharma Solutions is a world leader in organic-phase coacervation for pharmaceutical products.
Our proprietary taste masking platforms can improve product acceptability and patient adherence
Increasingly, format flexibility is becoming a requirement to address patient issues such as swallowing. The FDA also continues to push for pediatric-friendly formulations. Taste, smell, and mouth feel are the key product features to address when developing pediatric formulations. And Incremental formulation and clinical research costs require cost-effective solutions with flexibility in manufacturing scale. These drivers are why customers continue to come back to Adare Pharma Solutions for their taste-masking challenges.

 Among various taste masking strategies, physical barrier is one of the most efficient technologies, and Adare offers industrially feasible physical barrier technologies, such as Microcaps® and Optimµm®, for taste masking and dosage forms for pediatric populations.
Among various taste masking strategies, physical barrier is one of the most efficient technologies, and Adare offers industrially feasible physical barrier technologies, such as Microcaps® and Optimµm®, for taste masking and dosage forms for pediatric populations.
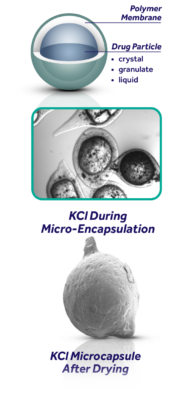
Microencapsulation by coacervation:
As the world leader in coacervation, Adare’s Microcaps® technology is widely scene as the industry leading taste-masking solution with a wide variety of delivery formats.
|
 |
|
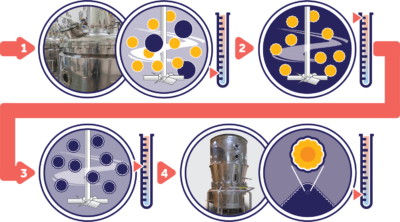
Proprietary Taste-Masking ProcessAlthough spherical particles are shown (yellow), the Microcaps® coacervation process can also be used for varying non-isometric particle shapes. Our Process Also Offers:
|
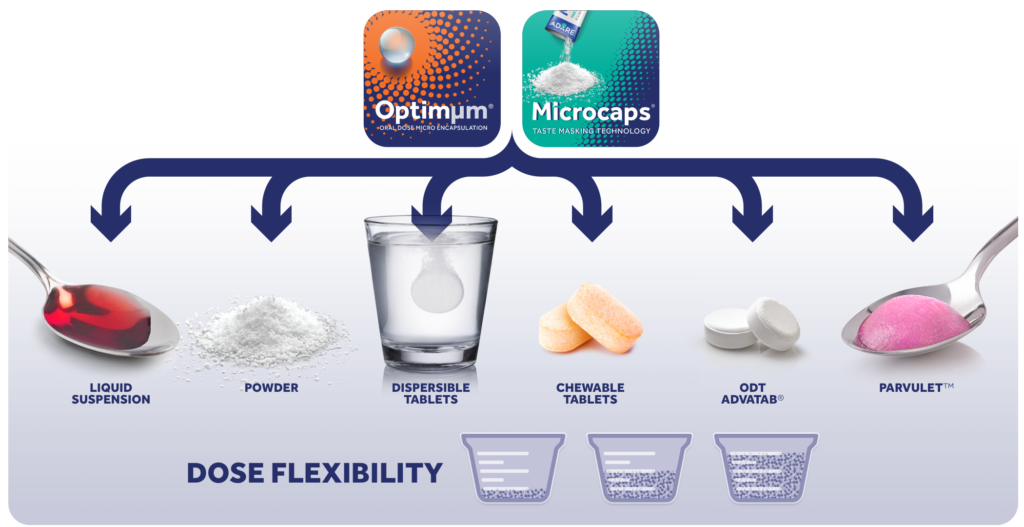
Adare’s Optimµm® technology provides innovative taste masking opportunities not seen before in oral pharmaceutical products
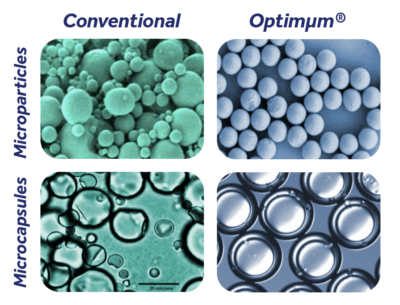
Optimµm® is developed and manufactured through Adare’s Precision Particle Fabrication™ technology, which produces uniform microspheres and microcapsules. Optimµm® is an elegant and robust solution with countless applications. You can learn more about how Optimµm® is manufactured using Precision Particle Fabrication™, here.
Optimµm® Multiparticulates are able to deliver:
- Uniformity
- Format Flexibility
- Dose Flexibility
- Taste Masking
- Variable Release Kinetics
- Single-step Microcapsules
The Broadest Variety of Taste-Masking Solutions in the Industry
Adare Pharma Solutions employs a variety of strategies for achieving successful taste-masking results for our customers, including:
API Taste Modifiers or Suppressants
API Solubility Modifier
|
API Barrier
API Modification Modify solubility or taste using:
|
Speak With An Expert

Nathan Dormer
Director, Drug Product Development
Nathan Dormer, Ph.D., is the Director of Drug Product Development at Adare Pharma Solutions and is responsible for pharmaceutical development activities at Adare’s Lenexa, KS and Philadelphia, PA facilities, where he supports a team of formulation scientists and analysts. He is a pharmaceutical scientist and bioengineer by training, with more than 16 years of experience developing microsphere-based solid oral and parenteral/implantable dosage forms in academic, innovator, and CDMO settings. Dr. Dormer earned his B.S. in Chemical Engineering and his Ph.D. (w/Honors) in Bioengineering from The University of Kansas while receiving NIH-sponsored Pharmaceutical Biotech Training.